Jan 02, 2015 The smallest serial dilution process is three 1.00 to 10.0 dilution steps. This dilution sequence results in about twice the measurement uncertainty. However, three 5.00 to 50.0 dilution steps or three 10.00 to 100.0 dilution steps could also be used to achieve the same dilution. Sep 28, 2009 A dilution in chemistry is a process that reduces the concentration of a substance in a solution. A serial dilution is the repeated dilution of a solution to amplify the dilution factor quickly. It’s commonly performed in experiments.
Parallel dilution is the dilution of a solution with equalquantity of the same solvent with which the solution is made. e.g.,1mL of 100µg/ml strength aqueous solution can be diluted to 2mL of50µg/mL strength solution by adding 1mL Water.
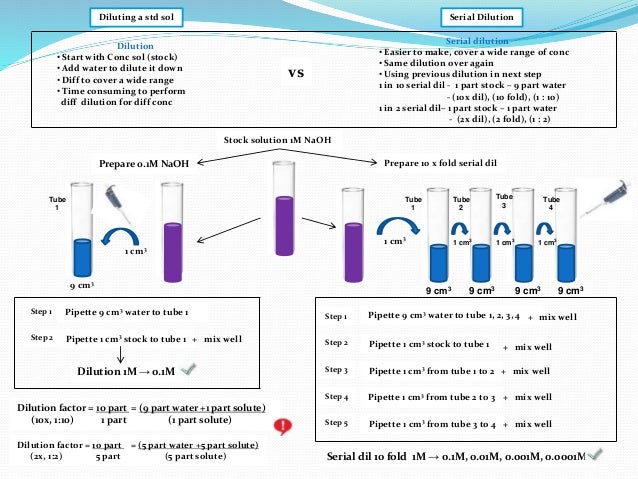
Definition of dilution and dilution factor?
As I understand it, the dilution refers to the dilution ratio. If you add 1 part of something to 4 parts of something else, the dilution ratio is 1 to 4. The dilution factor counts all the parts and expresses the same thing as 1 out of 5.

What is the effect of dilution on viscosity of oil?
dilution will reduce the viscosity The effect of dilution on viscosity of oil is that it will decrease.
12 Distinguish between dilution and dilution factor?
As I understand it, the dilution refers to the dilution ratio. If you add 1 part of something to 4 parts of something else, the dilution ratio is 1 to 4. The dilution factor counts all the parts and expresses the same thing as 1 out of 5.
What does 0 percent dilution mean?
What is the equation for a dilution?
What is infinite dilution?
Infinite dilution means such a large dilution so that when you add more solvent there is no change in concentration.
Distinguish between dilution and dilution factor?
Dilution is the process of making a less concentrated solution and dilution factor is the amount of solvent required to make a certain concentration of solution.
How does the number of moles of solute before a dilution compare with the number of solute after the dilution?
Before the dilution the number of moles is higher.
What is dilution factor?
In chemistry and biology, the dilution factor is the total number of unit volumes in which the material is dissolved. As I understand it, the dilution refers to the dilution ratio. If you add 1 part of something to 4 parts of something else, the dilution ratio is 1 to 4. The dilution factor counts all the parts and expresses the same thing as 1 out of 5.
What is the difference between dilution and diffusion?
what is the difference between a diffusion and a dilution
3 Fold Serial Dilution
How does dilution affect concentration of a solution?
Dilution reduces the concentration of a solution.
What is another term for dilution?
Dilution may also be described as 'watering down' or 'mixing', as well as 'diffusing', although none are exact synonyms of dilution.
What is helium dilution technique?
How is FRC measured by helium dilution, in specific detail.
What is the key to remember when considering the dilution of a solution?
To remember that only water is added in the dilution.
